

The general properties of metals can be summarised in the image below:
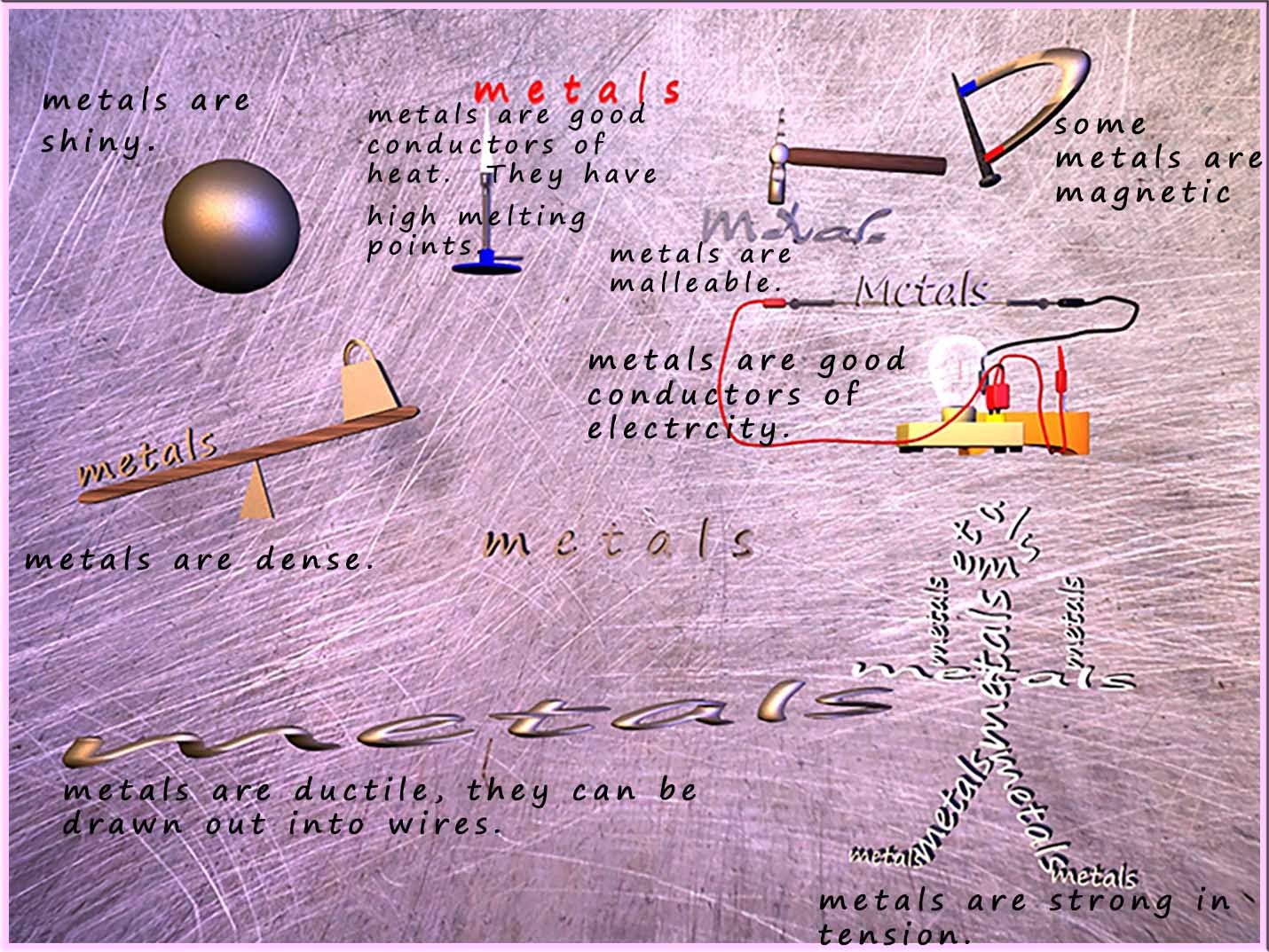
We can use the general properties of metals shown in the image above to help us figure out what the structure of a metal looks like. The table below tries to offer an explanation for some of the properties of metals and relate this property to a possible feature of the metal structure and the bonding present in the metal
| Metal property | How this relates to its structure |
|---|---|
| Metals are good conductors of electricity and heat | There must be free or delocalised electrons within the structure to conduct the electricity and heat and the density of these electrons within the lattice structure will strongly influence these properties. |
| Metals have high melting and boiling points. | Metals must have a giant structure with lots of strong bonds. |
| Metals are malleable (can be hammered into shape) and ductile (can be pulled into wire). | There are layers of particles that are able to slide over each other due to the non-directional nature of metallic bonds. |
| Metals are shiny. | Free or delocalised electrons are able to reflect light at the surface, this is simply because because these electrons can absorb and re-emit photons of light. |
| Metals are hard and dense | The ions present within the metal structure are packed tightly together; this tight packing of ions in a metallic lattice contributes to the high densities and the strong metallic bonds contribute to the hardness. |
These basic properties of metals allows us to suggest that
metals consist of a giant structures of ions
which are surrounded by a sea of free moving delocalised electrons.
We also already know that metals are found in the middle block of the periodic table and on the left hand side of the
periodic table in groups 1, 2 and 3 and that
they tend to lose electrons in their reactions and form
positively charged ions.
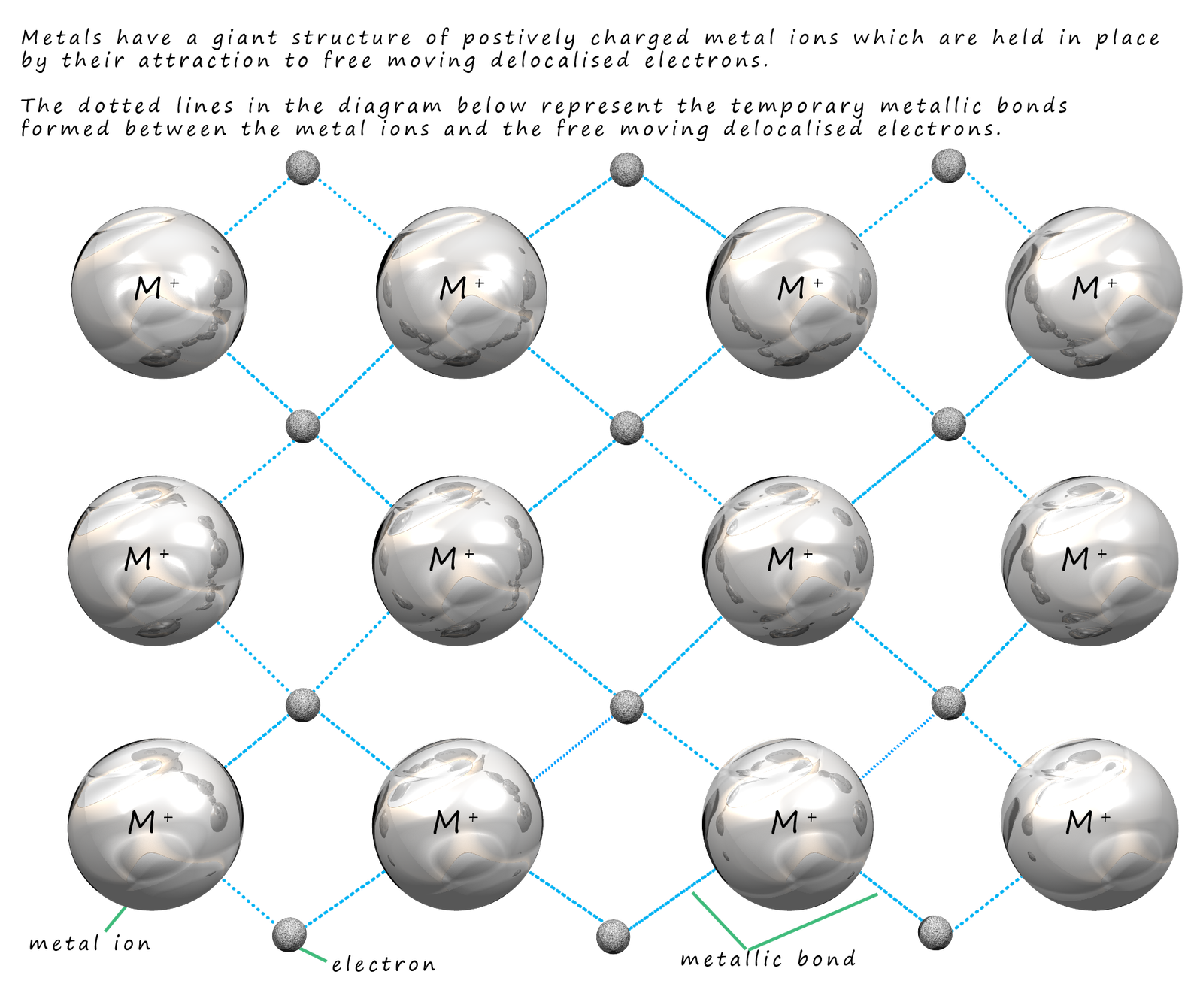
This leads us to the image shown below, here the dark grey balls represent the
positively charged metal ions (atoms which have
lost their outer shell electrons). These
electrons are delocalised and free to move through the
giant structure of ions.
This might seem odd since we might expect a giant structure of ions
which all have a positive charge to repel each other and so
the structure would simply break down. However we need to think about
the delocalised electrons within the structure.

The negatively charged electrons are attracted to any neighbouring
positively charged metal ions and
this prevents the metal ions from
repelling
each other. This attraction of the negatively
charged electrons to the neighbouring positively charged
metal ions is called a metallic
bond, a metallic bond is the electrostatic attraction between the positively charged metal cations and the sea of delocalised electrons. A key feature of
this bond is the fact that metallic bonds are not permanent but are
able to break and reform
as the electrons can move
freely around the structure, this is shown in the diagram below:
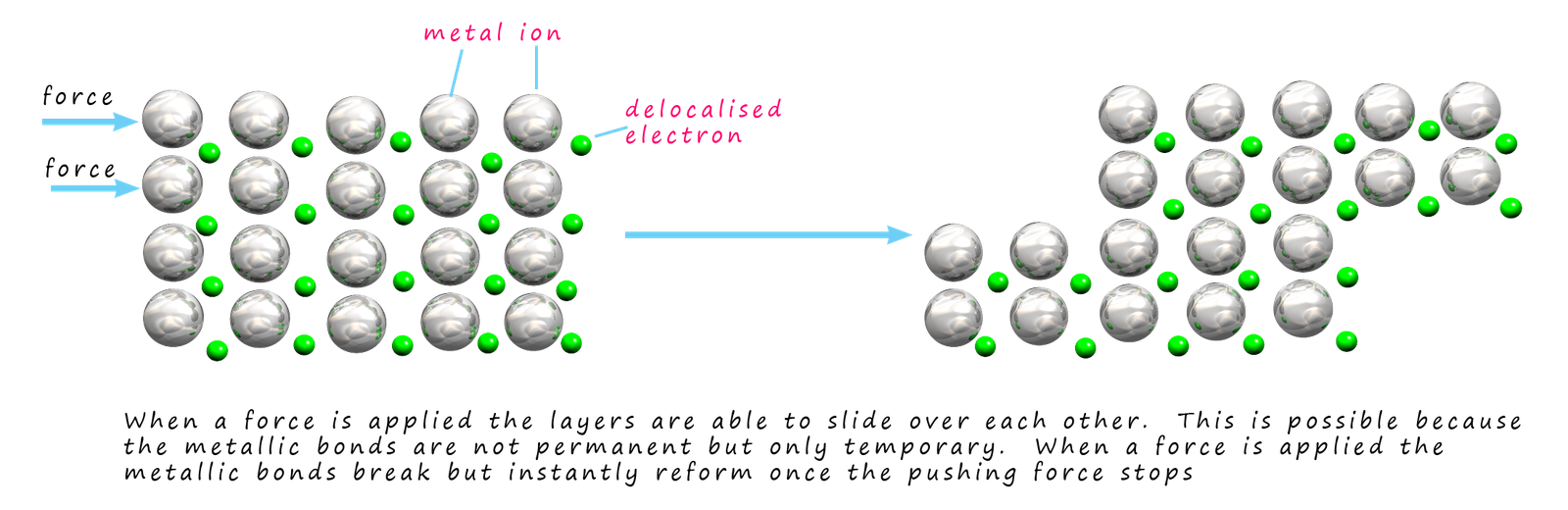
The fact that these metallic bonds are not permanent
but are able to break and
reform allows the layers of ions
to slide over each other if a large enough force is applied to them. This is shown in the diagram below; which shows
a pushing force being applied to the top two
layers of ions in a metal structure.
The metallic bonds in these layers immediately break
and the layers slide along
but as soon as they stop moving the metallic bonds immediately reform.
This is would explain why metals are malleable (can be hammered into shape) and ductile (can be pulled into wires).
Metals generally have high melting points, high densities and are good electrical and thermal conductors, though these properties can vary significantly as we cross the periodic table. Let us examine some these typical properties of metals in a little more detail